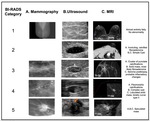
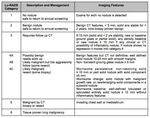
1.- BI-RADS (Table 1,
Figure 1)
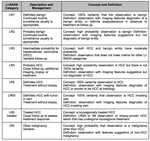
The Breast Imaging Reporting and Data System(BI-RADS)was developed by the American College of Radiology to serve as a guide providing standarized breast imaging terminology,
a recommended reporting structure including final assessment categories with accompanying management recommendations,
and a framework for data collection and auditing.
Subsequent validation showed that implementation of BI-RADS reduces inter and intraobserver variability [2].
The first edition was released in 1993 for mammography findings,
since then more editions have been released.
In 2003 the first edition of the ultrasound and magnetic resonance imaging BIRADS atlas was published [3].
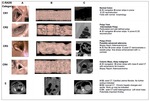
2.- C-RADS (Table 2,
Figure 2)
The Working Group on Virtual Colonoscopy published in 2005 a guide to the interpretation of computed tomography (CT) colonography results: the CT Colonography Reporting and Data System,
or “C-RADS.” It includes a description of terms for reporting the size,
morphologic features,
and location of polyps and masses,
a description of a reporting scheme for colonic and extracolonic lesions with management recommendations [4].
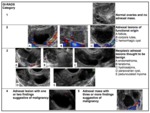
3.- GI-RADS (Table 3,
Figure 3)
Amor et al.
in 2009 described a reporting system called the Gynecologic Imaging Reporting and Data System (GI-RADS) for reporting findings in adnexal masses based on transvaginal sonography and providing an estimated risk of malignancy according to this classification. The proposed reporting system showed good diagnostic performance and the interobserver agreement was high [5,6].
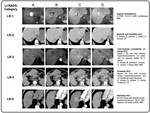
4.- LI-RADS (Table 4,
Figure 4)
In 2008,
the American College of Radiology developed a system for standardized interpretation,
reporting,
and data collection for CT and MRI examinations in patients at risk for HCC.
Version 1.0 of the resulting Liver Imaging Reporting and Data System (LI-RADS) was released in 2011,
the version 2013,
includes a lexicon and an imaging atlas [7].
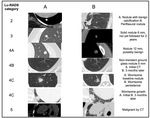
5.- Lu-RADS (Table 5,
Figure 5)
In the 98th Scientific Assembly and Annual Meeting of Radiological Society of North America,
Manos et al.
introduced the Lung Reporting and Data System (LuRADS) for CT- screening detected lung nodules to guide management,
facilitate communication and provide a framework of data collection and analysis [8].
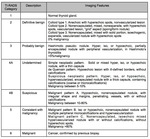
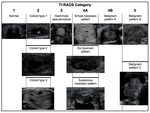
6.- TI-RADS (Table 6,
Figure 6)
There has been suggested several thyroid imaging reporting and data systems since 2009.
Among these systems there is no consensus concerning the exact format and style for reporting the results of thyroid ultrasound [9-12].
Horvath et alestablished 6 categories and called it Thyroid Imaging Reporting and Data System (TI-RADS) based on 10 sonographic patterns.
They suggested that patients with TI-RADS 4 and 5 nodules must be biopsied [9].Interobserver agreement with this classification has been reported fair to moderate,
with a high negative predicitive value [13,14].
7.- PI-RADS (Table 7,
Figure 7)
The European Society of Urogenital Radiology (ESUR) released in 2012 a standardized system for prostate magnetic resonance imaging (MRI) interpretation and reporting of results: Prostate Imaging Reporting and Data System(PI-RADS).
In this scoring system every parameter: T2WI,
DWI,
DCE-MRI and MRSI (optional) is scored on a five-point scale, Additionally,
each lesion is given an overall score,
to predict its chance of being a clinically significant cancer,
the scoring should include as a minimum requirement division of the prostate 16 regions,
as an optimal requirement into 27 regions.
[15]
It has been reported good to moderate inter-reader agreement for the PI-RADS score [16,
17].
In the study by Schimmöller et al.
[16] PI-RADS showed high sensitivity and negative predictive value for biopsied lesions.
Roethke et al.
evaluated the PI-RADS for detection of prostate cancer in patients with magnetic resonance/transrectal ultrasound fusion-guided biopsy,
they concluded the system is beneficial to indicate the likelihood of cancer and it is also valuable to identify locations to be targeted with biopsy [18].